 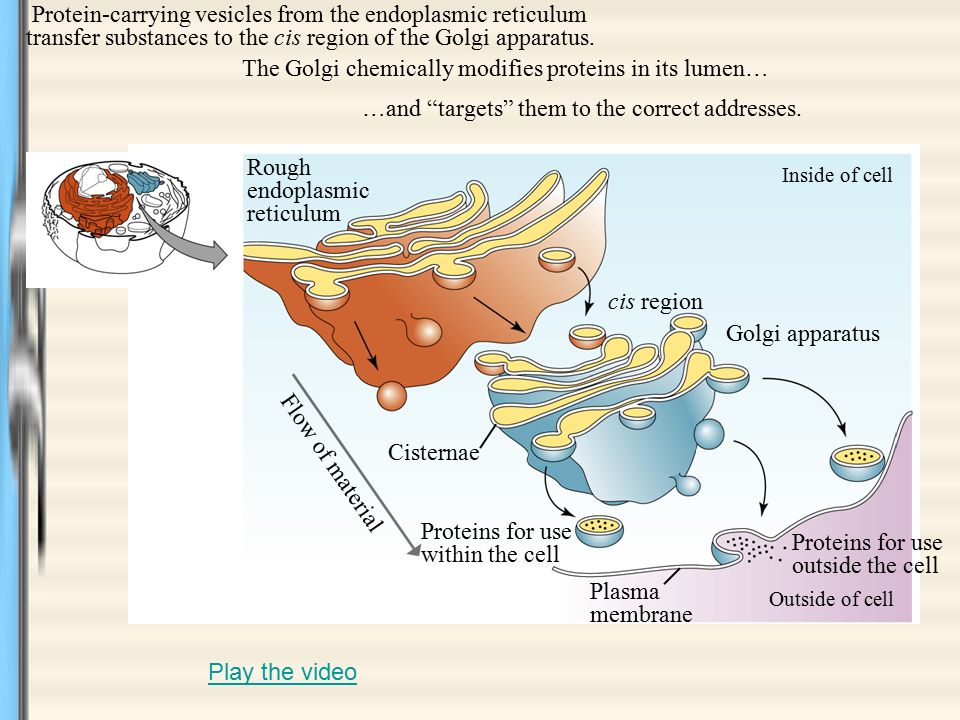
The growing polypeptide emerges on the lumenal side of the membrane and begins to fold in the ER lumen. Thus, the current model of protein translocation involves a multiprotein pore, termed the translocon pore, with an aqueous channel of 9 to 15 Å in diameter when inactive and 40 to 60 Å when operational ( Hamman et al., 1998). Studies with fluorescently labeled proteins, however, have shown that polypeptides remain in an aqueous environment during translocation ( Crowley et al., 1993). Removal is performed by signal peptidase, an enzyme located on the lumenal surface of the ER membrane.įor many years, translocation across the ER membrane was thought to occur directly through the membrane itself by virtue of the hydrophobic core of the signal peptide. The N-terminal signal peptide is removed cotranslationally while the nascent polypeptide is emerging into the ER lumen ( Vitale et al., 1993). Integral membrane proteins may also be synthesized with an N-terminal signal peptide, but their orientation within the ER membrane and the translocation of portions that will eventually reside in the lumen are determined by the corresponding membrane-spanning domains. Translocation of soluble proteins depends on the N-terminal signal peptide. 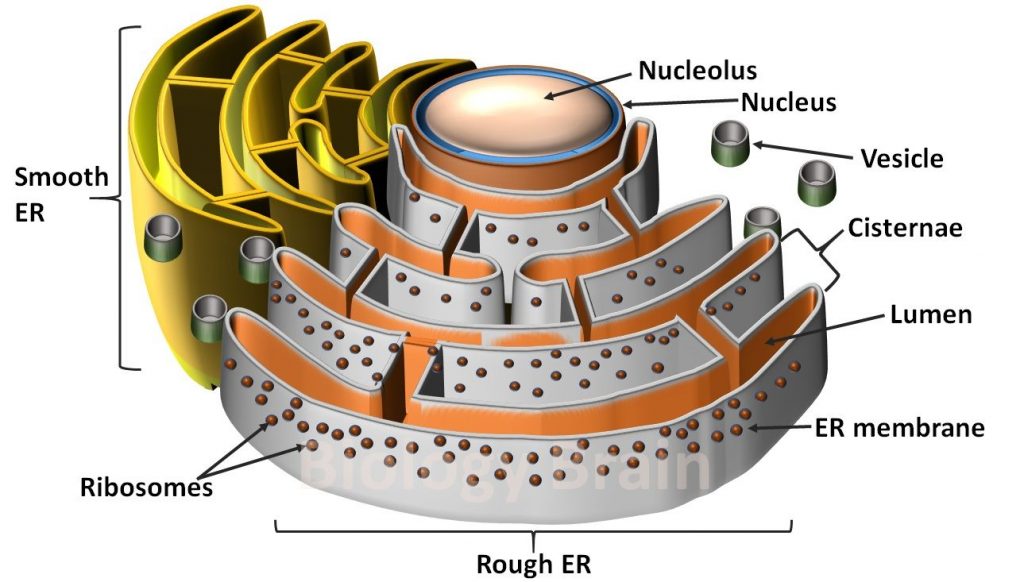
Secretory proteins usually enter the ER cotranslationally. These safeguards not only optimize folding and assembly of newly synthesized secretory proteins but also dispose of defective ones ( Haas and Wabl, 1983 Munro and Pelham, 1986 Hammond et al., 1994 Wiertz et al., 1996 Pedrazzini et al., 1997). The crucial role of the ER in safeguarding the correct folding and assembly of proteins has become clearer in the past two decades with the discovery of the ER molecular chaperone machinery and the associated quality control mechanisms (the term “quality control” was introduced in a review by Hurtley and Helenius, 1989). It takes care of the folding and assembly not only of its own residents but also of proteins destined to other locations. The ER is therefore unique among the compartments of the eukaryotic cell. However, the mechanisms of traffic along the secretory pathway involve only a single translocation event across the ER membrane, after which proteins do not have to cross any further membrane to reach other stations along the pathway. Because nearly all eukaryotic proteins are synthesized in the cytosol, those destined to reside in a different location must cross membranes in an unfolded state and then fold and assemble in the compartment of destination. The lumens of the ER, Golgi complex, and vacuole are topologically equivalent to the cell exterior, and extracellular domains of plasma membrane proteins are thus lumenal when they start their journey in the ER. Protein trafficking in the secretory pathway can occur both via vesicle budding with subsequent fusion and via passage along connecting tubules. Routes from the ER to vacuoles, bypassing the Golgi complex, also exist ( Levanony et al., 1992 Robinson et al., 1995 Hara-Nishimura et al., 1998 Jiang and Rogers, 1998 see Sanderfoot and Raikhel, 1999, in this issue). The retrograde traffic allows for endocytosis of extracellular molecules as well as recycling of membranes and proteins to maintain the integrity of the different compartments. 
This biosynthetic, or anterograde, traffic is balanced by retrograde traffic running in the opposite direction. In the secretory pathway, proteins travel from the ER through the Golgi apparatus to arrive at the cell surface or at vacuoles. A number of intermediate compartments have been identified more recently (see Battey et al., 1999 Marty, 1999 Sanderfoot and Raikhel, 1999, in this issue). In a review based mainly on observations of plant cells, Morré and Mollenhauer ( 1974) posited the endomembrane system as the functional integration of the ER, Golgi complex, secretory vesicles, plasma membrane, and hydrolytic compartments (vacuoles in plants and lysosomes in animals). In the mid to late 1960s, the discovery that eukaryotic secreted proteins are first segregated in the lumen of the endoplasmic reticulum (ER) before traveling within membranous structures to reach the cell surface placed the ER at the start point of a newly recognized metabolic pathway now known as the secretory pathway ( Palade, 1975). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
